Publications
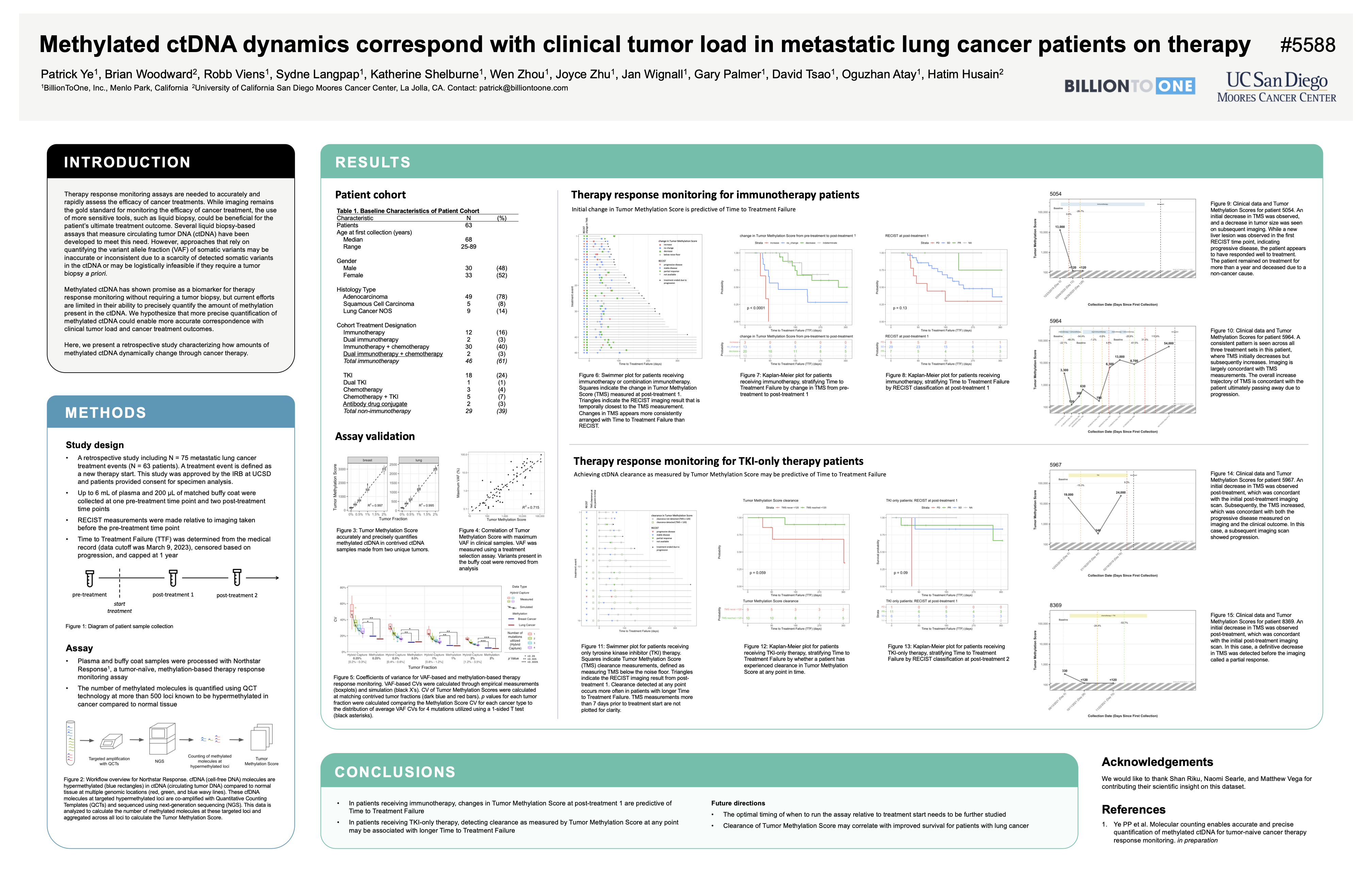
Longitudinal Clinical Validation of the Northstar Response Using Methylated cDNA as a Dynamic Treatment Response Biomarker in Advanced Stage Gastrointestinal (GI) Cancers
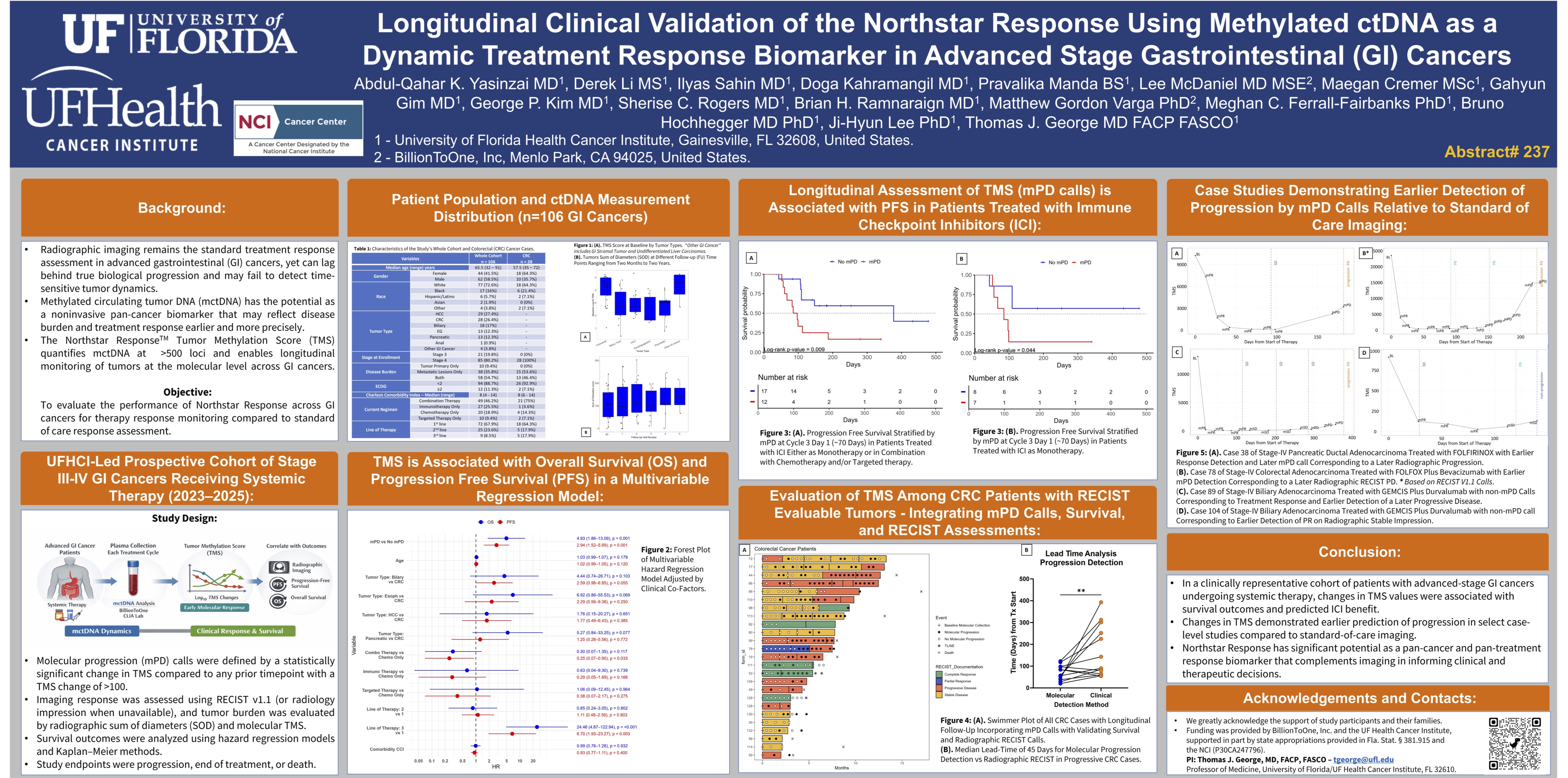
Longitudinal Clinical Validation of the Northstar Response Using Methylated cDNA as a Dynamic Treatment Response Biomarker in Advanced Stage Gastrointestinal (GI) Cancers

Liquid biopsy to reveal colorectal cancer molecular subtype information from ctDNA epigenetics

Liquid biopsy to reveal colorectal cancer molecular subtype information from ctDNA epigenetics

Quantifying MLH1 promoter methylation via liquid biopsy
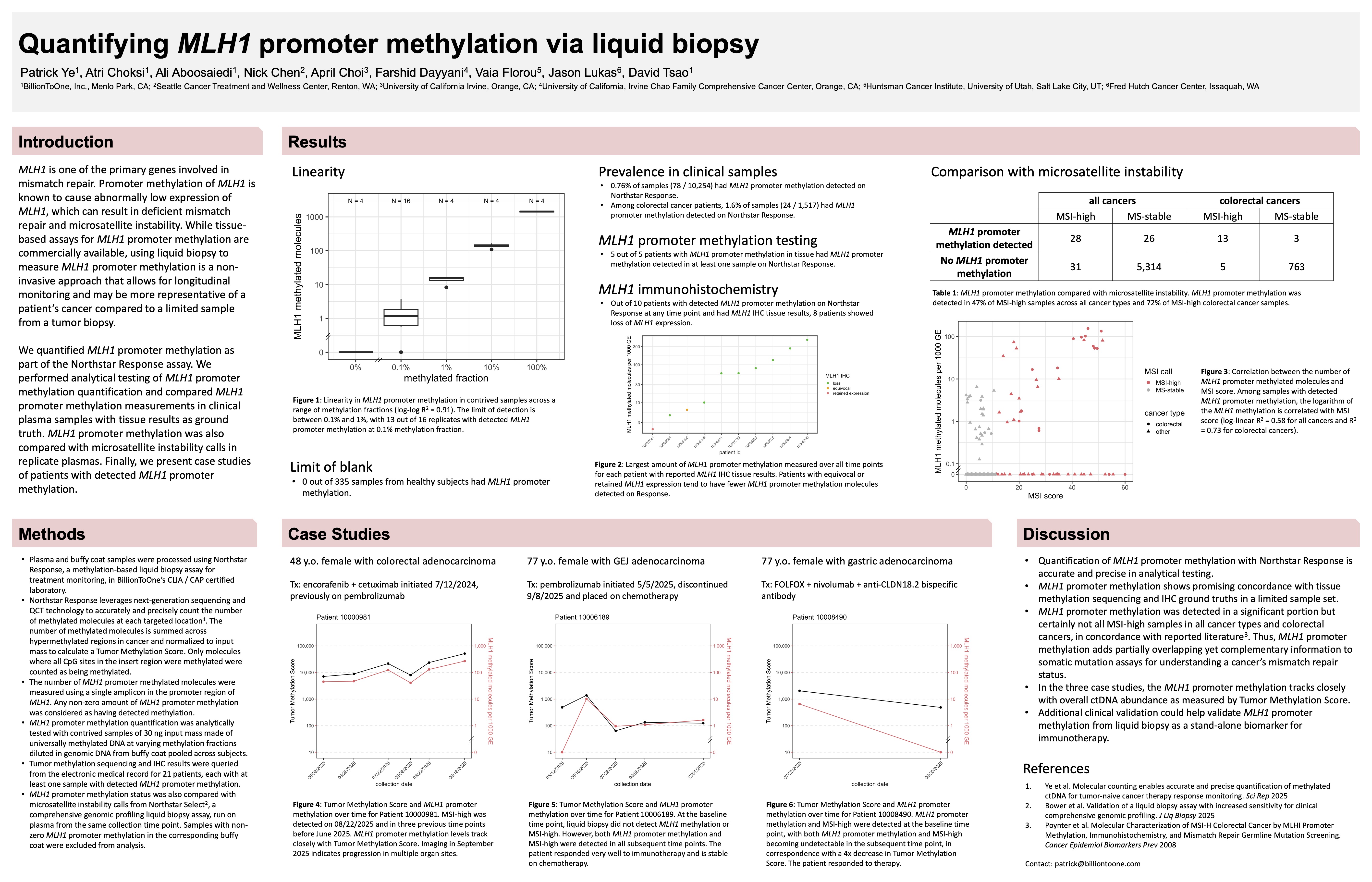
Quantifying MLH1 promoter methylation via liquid biopsy

Prevalence and characterization of ESR1 alterations detected with an ultra-sensitive, liquid-only CGP assay in a large breast cancer cohort
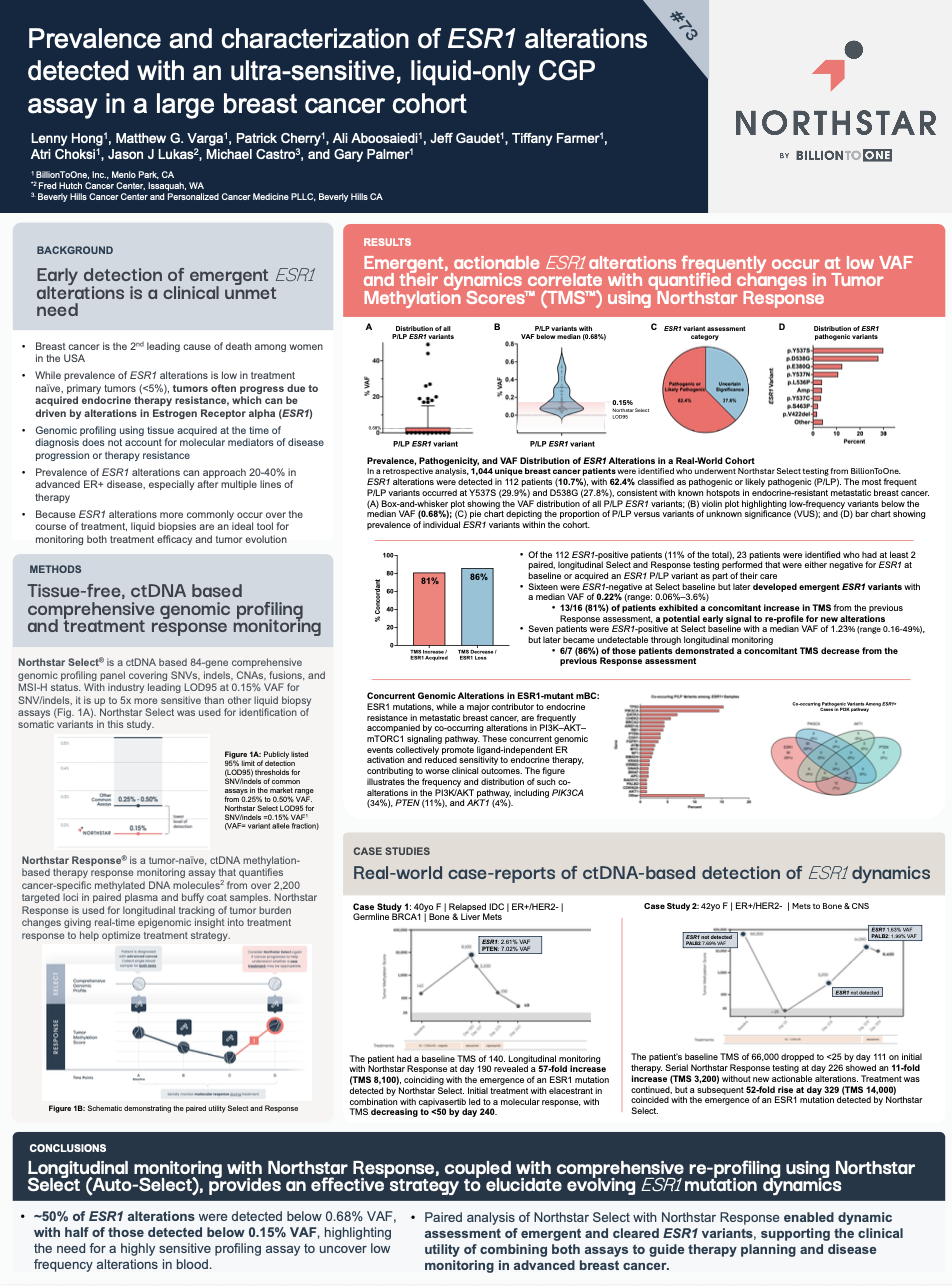
Prevalence and characterization of ESR1 alterations detected with an ultra-sensitive, liquid-only CGP assay in a large breast cancer cohort

Validation of a liquid biopsy assay with increased sensitivity for clinical comprehensive genomic profiling

Validation of a liquid biopsy assay with increased sensitivity for clinical comprehensive genomic profiling

Circulating cell-free methylated tumor DNA measurements correlate with plasma VAF-based tumor fraction estimates

Circulating cell-free methylated tumor DNA measurements correlate with plasma VAF-based tumor fraction estimates

Detection of a novel GNA11 processed pseudogene in chromosome 20 from cfDNA and implications for liquid biopsy

Detection of a novel GNA11 processed pseudogene in chromosome 20 from cfDNA and implications for liquid biopsy

Tumor fraction estimation and tissue copy number inference using copy number signal from a liquid biopsy assay

Tumor fraction estimation and tissue copy number inference using copy number signal from a liquid biopsy assay

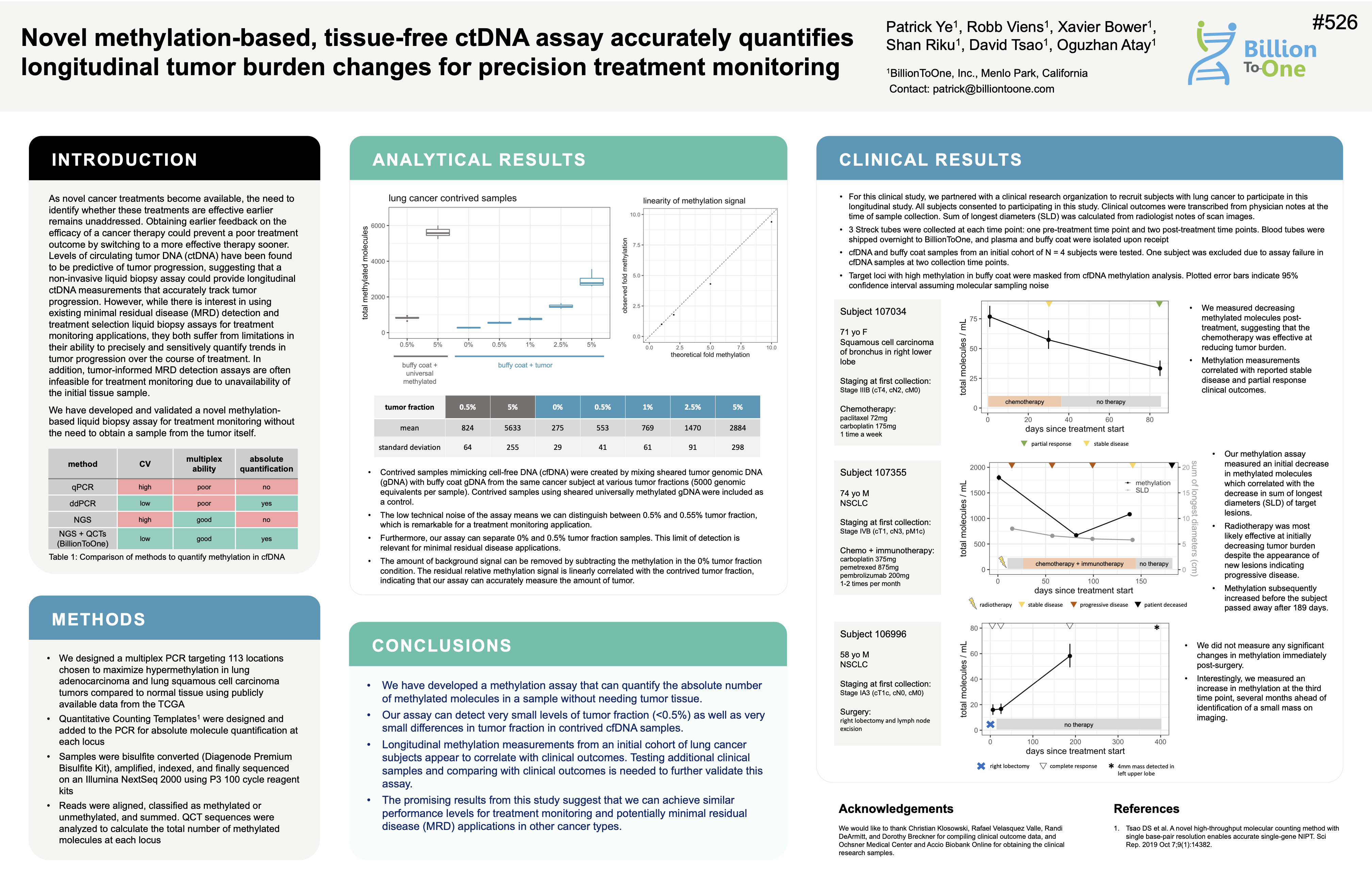
Molecular counting enables accurate and precise quantification of methylated ctDNA for tumor-naive cancer therapy response monitoring

Molecular counting enables accurate and precise quantification of methylated ctDNA for tumor-naive cancer therapy response monitoring

Clinical Validation of the Northstar Response: A Novel Quantitative Methylation ctDNA Monitoring Assay for Advanced GI Cancer Treatment Response
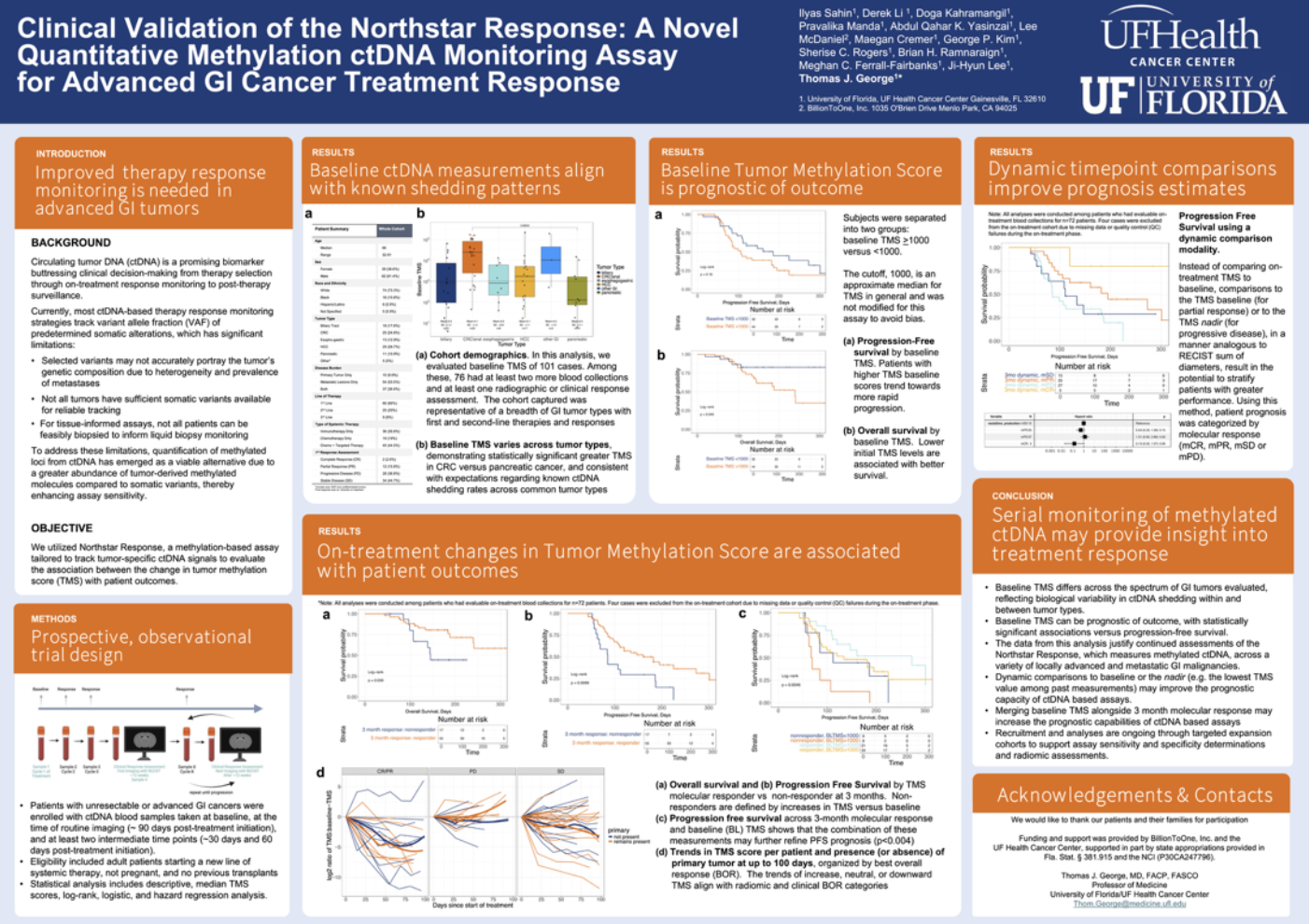
Clinical Validation of the Northstar Response: A Novel Quantitative Methylation ctDNA Monitoring Assay for Advanced GI Cancer Treatment Response

Absolute quantification of cell-free DNA for prenatal genetics and oncology

Absolute quantification of cell-free DNA for prenatal genetics and oncology

Brief Report: Methylation-Based ctDNA Serial Monitoring Correlates With Immunotherapy Response in NSCLC

Brief Report: Methylation-Based ctDNA Serial Monitoring Correlates With Immunotherapy Response in NSCLC

Methylation-Based ctDNA Serial Monitoring Correlates with Immunotherapy Response in NSCLC

Methylation-Based ctDNA Serial Monitoring Correlates with Immunotherapy Response in NSCLC

A novel liquid biopsy assay with an 88% detection rate of genomic alterations across CNS tumors.
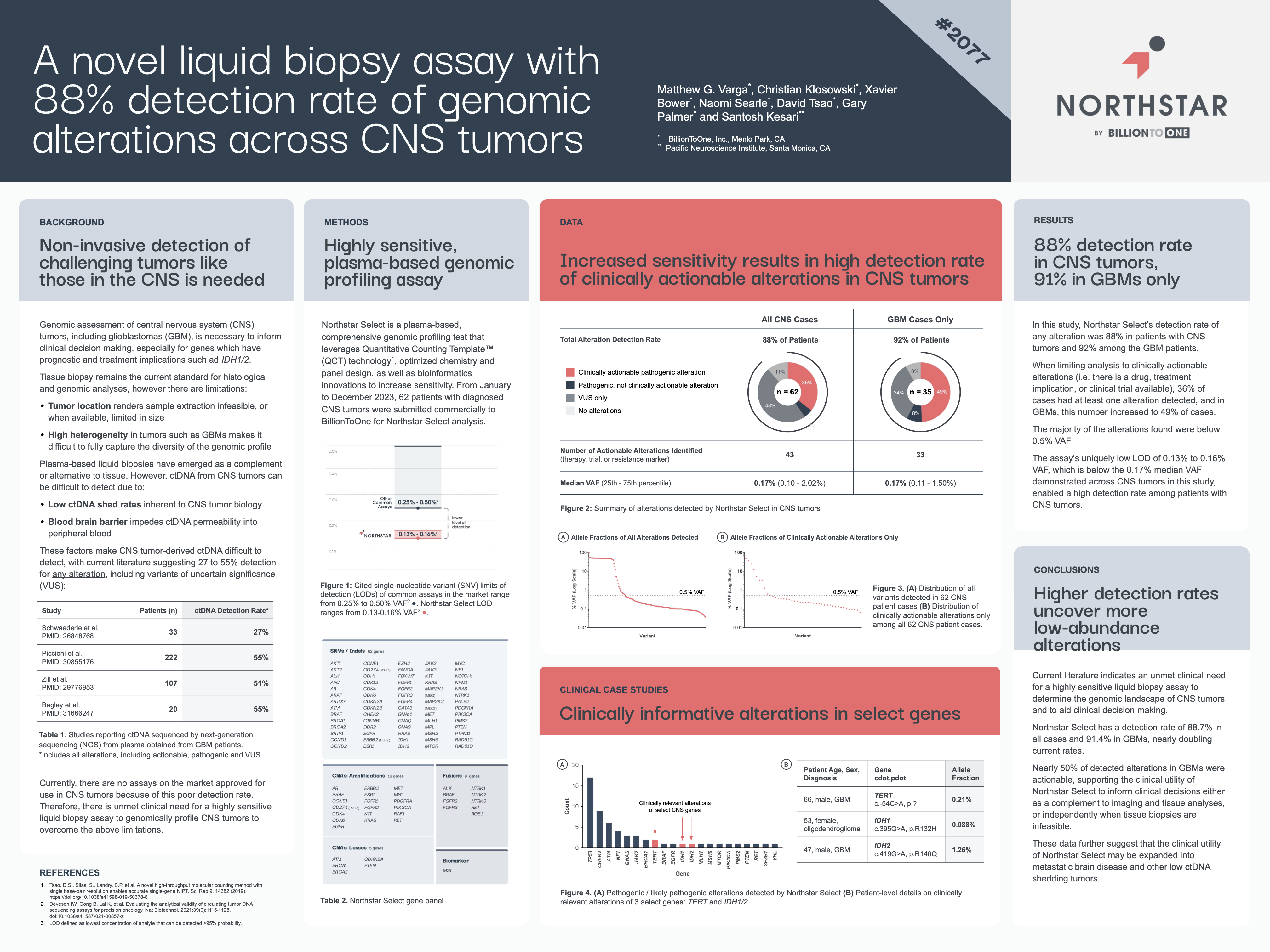
A novel liquid biopsy assay with an 88% detection rate of genomic alterations across CNS tumors.

Clinical validation of Northstar Select, a novel liquid biopsy assay for comprehensive genomic profiling of solid tumors.
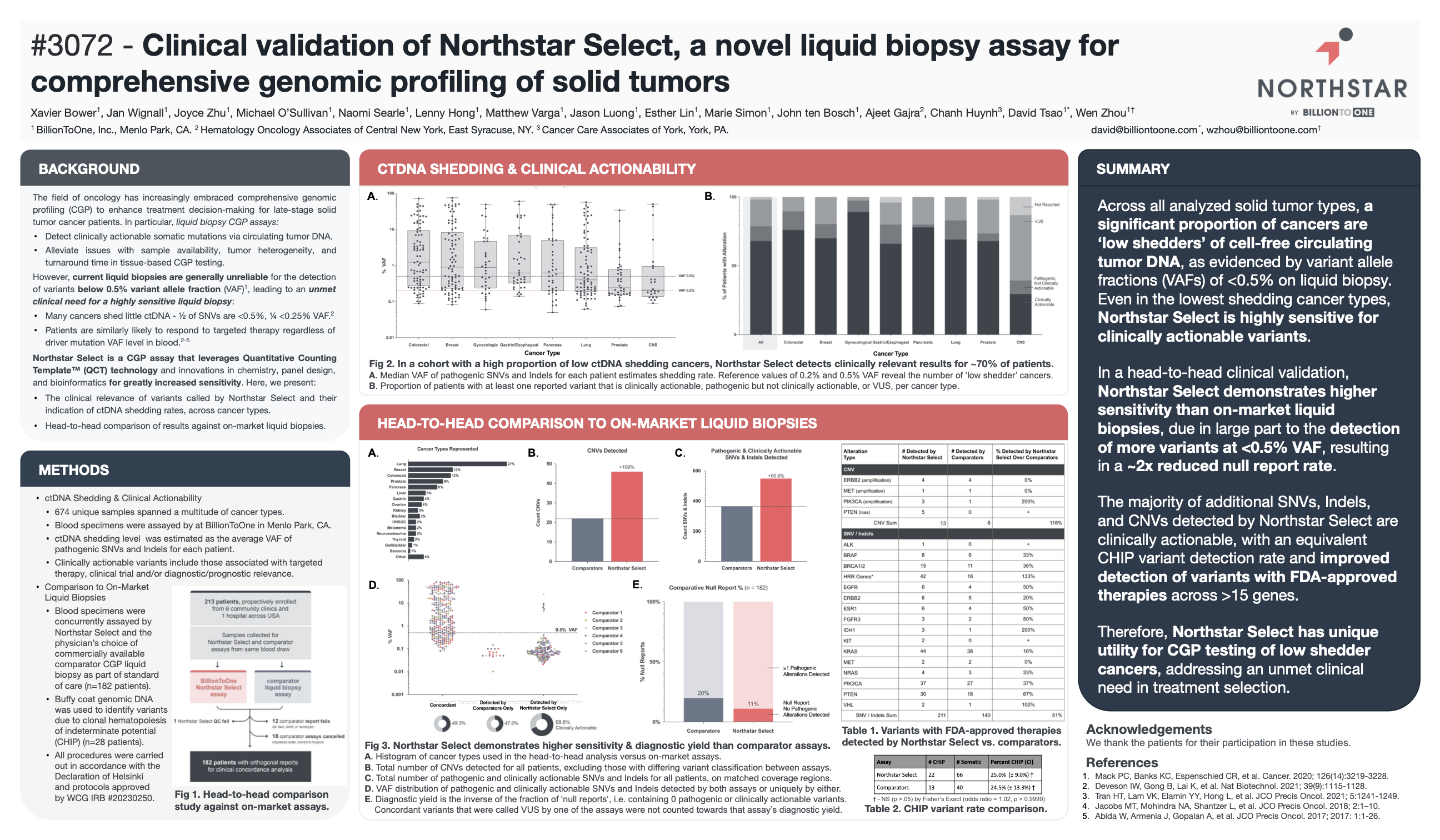
Clinical validation of Northstar Select, a novel liquid biopsy assay for comprehensive genomic profiling of solid tumors.

Analytical and clinical validity of Northstar Select, a quantitatively enhanced liquid biopsy assay for comprehensive genomic profiling of solid tumors

Analytical and clinical validity of Northstar Select, a quantitatively enhanced liquid biopsy assay for comprehensive genomic profiling of solid tumors

Clinical Validation of Northstar Response, a novel methylated ctDNA therapy response monitoring assay in patients with advanced GI cancer undergoing active treatment

Clinical Validation of Northstar Response, a novel methylated ctDNA therapy response monitoring assay in patients with advanced GI cancer undergoing active treatment

Methylated ctDNA as a biomarker for detecting previously undetectable clinically significant somatic mutations.

Methylated ctDNA as a biomarker for detecting previously undetectable clinically significant somatic mutations.